Introduction: The Quiet Crisis Inside Every Hospital
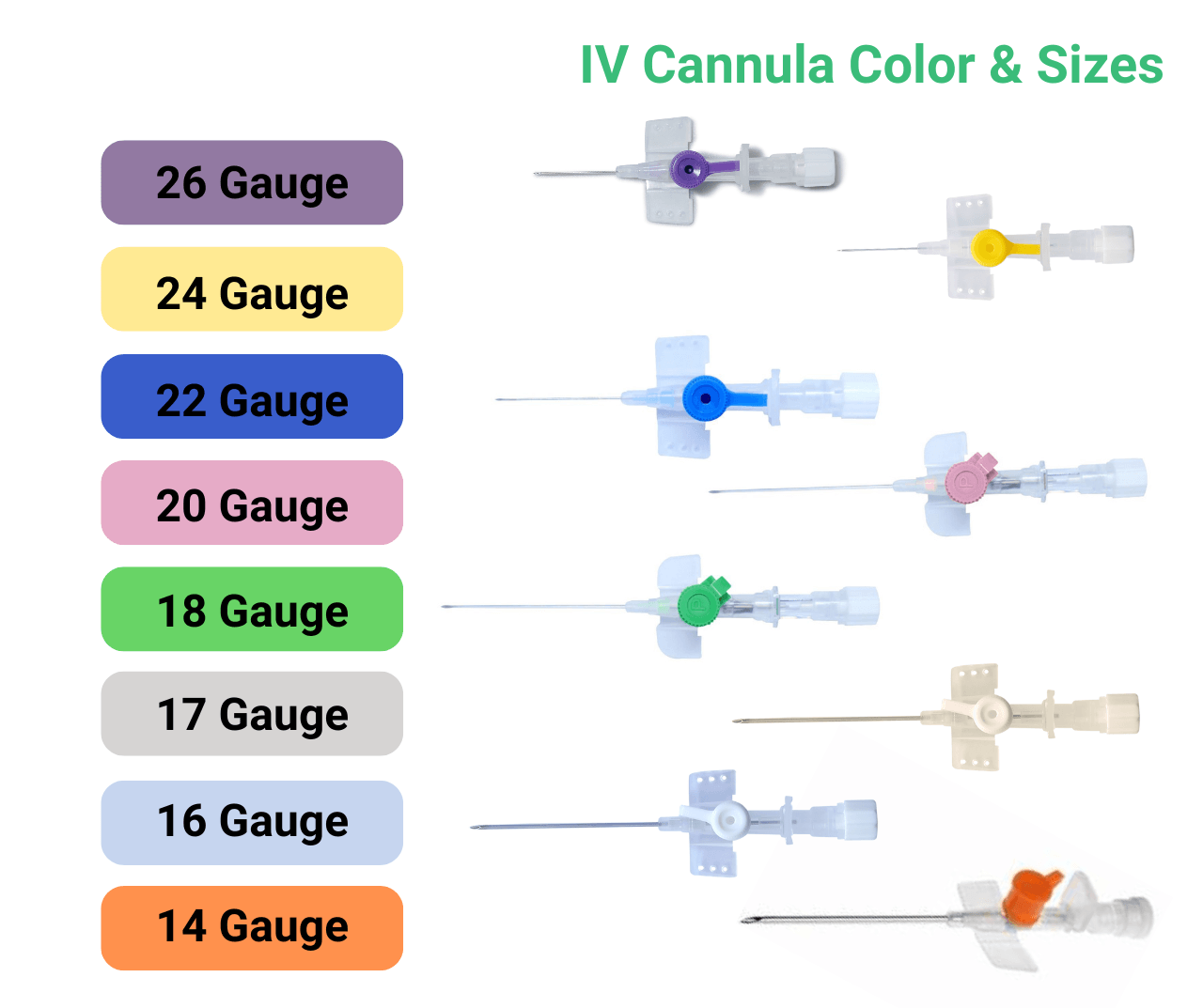
Walk into any hospital ward today, and you’ll find IV cannulas in nearly every patient’s hand. Globally, over one billion peripheral intravenous catheters (PIVCs) are inserted every year, making them the most common invasive medical device in clinical practice. Studies show that around 90% of hospitalized patients receive some form of IV therapy during their stay.
Yet behind this routine procedure lies a problem that has stubbornly resisted decades of attention: IV infiltration and extravasation. When fluids or medications leak out of the vein into surrounding tissue, the consequences range from mild swelling to permanent tissue damage, compartment syndrome, and even amputation in the worst cases.
Clinical research has shown that infiltration is the single most common PIVC complication, with one major study reporting an incidence of nearly 18% in hospitalized adults. In paediatric populations, the numbers climb higher still.
Now, a new generation of technology, AI-powered smart cannulas, promises something the industry has chased for years: the ability to detect infiltration before it causes harm.
Section 1: The Scale of the Infiltration Problem
Before exploring the technology, it’s worth understanding why this matters so much to hospital administrators, clinicians, and procurement leaders.
The Clinical Burden
IV infiltration occurs when fluid escapes the vein into the surrounding tissue. When the leaked fluid is a vesicant chemotherapy agent, vasopressors, calcium solutions, or high-concentration dextrose, the result is extravasation, which can cause:
- Tissue necrosis
- Nerve damage
- Compartment syndrome
- Permanent scarring or loss of limb function
- Surgical intervention, including skin grafts
Children, elderly patients, and oncology patients face the highest risk. A single severe extravasation event in a paediatric patient can lead to lifelong disability.
The Economic Burden
The financial picture is equally sobering:
- United States: Premature PIVC removal alone is conservatively estimated to cost hospitals $1.5 billion annually. Patients with PIVC complications had a mean hospitalisation cost of $10,895 versus $7,009 for those without, an adjusted difference of nearly $4,000 per patient.
- United Kingdom: Extravasation injury claims cost the NHS approximately £16 million between 2010 and 2021, with individual case costs reaching nearly £39,000.
- Australia: Annual cost of PIVC-related occlusion, infiltration, and dislodgement in adult emergency departments is estimated at $14 million AUD.
- China: Tertiary hospital studies show treatment costs scaling sharply with infiltration severity grade.
The global peripheral IV infiltration detection devices market was valued at $1.3 billion in 2025 and is projected to reach $3.7 billion by 2036, growing at a 9.9% CAGR. India is expected to lead growth at a 10.9% CAGR, driven by hospital infrastructure expansion and patient safety investment.
This is no longer a niche clinical concern, it is a regulatory, financial, and reputational priority for healthcare systems worldwide.
Section 2: Why Traditional Monitoring Falls Short
The current standard of care for monitoring IV sites relies almost entirely on visual inspection by nursing staff, typically every one to two hours. While well-intentioned, this approach has several inherent weaknesses:
- It is reactive, not predictive. By the time swelling, blanching, or coolness becomes visible, fluid has already escaped into the tissue.
- It depends on workload. Nursing shortages and high patient-to-nurse ratios mean inspection intervals are often longer in practice than in protocol.
- It misses early-stage infiltration. Subclinical leakage of small volumes is virtually impossible to detect by sight or touch.
- It varies by clinician. Subjective grading of infiltration severity introduces inconsistency in detection and intervention.
- It fails in covered sites. When the IV site is covered by a dressing, splint, or blanket, as is common in paediatric and post-surgical care, visual cues disappear entirely.
The result: infiltration is often caught late, when damage has already begun.
Section 3: The Rise of Smart Cannula Technology
Over the past five years, medical device research has converged on a powerful idea, embedding sensors directly into or alongside the IV cannula to create a continuous, intelligent monitoring system. These systems use a combination of hardware sensors and AI algorithms to detect infiltration in its earliest stages.
How Smart Cannulas Work
Modern smart cannula systems typically integrate one or more of the following sensing modalities:
1. Optical Sensors
Infrared or single-wavelength light is directed into the tissue near the IV site. The light reflected, scattered, or absorbed by the tissue is measured continuously. When fluid begins to accumulate outside the vein, the optical signature changes, and the system flags an alert. Patented optical detection systems compare real-time readings against a patient-specific baseline established at the time of cannula insertion.
2. Electrical Bioimpedance (EBI) Sensors
Tissue impedance changes as extravascular fluid accumulates. Wearable EBI sensors positioned around the insertion site measure these changes continuously. Multi-sensor systems pair EBI with motion sensors to filter out false positives caused by limb movement.
3. Pressure and Flow Sensors
By analysing the pressure waveform and flow signatures within the IV line itself, AI models can detect occlusions, partial dislodgement, and venous changes that precede infiltration.
4. Multi-Modal Sensor Fusion
The most advanced systems combine optical, impedance, temperature, skin strain, and motion data, feeding all streams into a machine learning model trained to distinguish true infiltration from artefacts.
Where AI Adds Real Value
A simple sensor can detect change. AI is what separates a true clinical alert from a false alarm. Machine learning models trained on large multi-patient datasets can:
- Establish personalised baselines that adapt to each patient’s tissue characteristics
- Distinguish infiltration signatures from movement artefacts, postural changes, or temperature fluctuations
- Predict the probability of infiltration developing in the next 30 to 60 minutes
- Learn from continuous monitoring data across thousands of patients to improve sensitivity and specificity over time
In a large retrospective study of an FDA-cleared continuous IV monitoring system, the use of optical AI-based monitoring alongside standard care resulted in a 93% reduction in injury severity. A financial impact model for the same technology projected annual margin improvements of $5.1 to $7.9 million for non-paediatric hospitals and up to $13.4 million for a 150-bed paediatric hospital, a return on investment of 7x to 14x.
Section 4: What This Means for Hospitals and Health Systems
For hospital administrators and procurement leaders, the question is no longer whether smart IV monitoring is coming; it is when and how to integrate it.
Regulatory Pressure Is Building
In the United States, CMS quality programmes are increasingly mandating infiltration detection as part of IV therapy safety protocols. The UK’s NHS is accelerating adoption through national procurement frameworks targeting paediatric and general ward settings. India’s National Health Mission and major private hospital chains are evaluating smart monitoring as part of tertiary-care safety upgrades.
Liability and Litigation Are Reshaping Decisions
Hospitals are no longer judged solely on whether infiltration happened, but on whether monitoring systems capable of catching it were in place. As one industry analyst put it, infiltration detection has shifted from a “bedside accessory decision” to a “patient safety and liability management investment”.
Workflow Integration Will Make or Break Adoption
Smart cannula technology will only succeed if it integrates seamlessly with:
- Existing electronic health records (EHRs)
- Nurse call systems and centralised monitoring dashboards
- Hospital Wi-Fi and IoT infrastructure
- Standardised IV therapy protocols
Vendors that deliver clinical accuracy and operational simplicity will dominate the next decade.
Section 5: The Manufacturer’s Perspective — A New Era for Medical Disposables
For medical disposable manufacturers, the rise of smart cannulas raises important strategic questions. The traditional IV cannula has been a high-volume, low-margin product, optimised for sterility, biocompatibility, and ease of insertion. Smart cannulas introduce a new dimension entirely: electronics, software, data, and connectivity, all in a single-use device.
This creates several opportunities:
- Hybrid product portfolios combining conventional cannulas (for routine use) with sensor-enabled variants (for high-risk patients).
- Partnerships with healthtech firms for sensor integration, algorithm development, and cloud platforms.
- Regulatory leadership — manufacturers with strong CE, FDA, ISO 13485, and CDSCO compliance frameworks will have a head start, since smart devices face stricter approval pathways.
- Sustainability innovation — smart, single-use electronics raise serious e-waste questions, opening doors for biodegradable substrates and recyclable sensor designs.
At Lars Medicare, we believe the future of IV therapy will be defined by three principles: clinical safety, intelligent design, and global accessibility. As we expand our portfolio of safety IV cannulas, Blood Control Technology (BCT) devices, and closed-loop IV systems, we continue to monitor and contribute to the evolution of smart, sensor-integrated IV therapy.
Section 6: What’s Next — The Five-Year Outlook
Looking ahead, several developments are likely to reshape the smart IV therapy landscape:
- Miniaturisation of sensors to the point where they can be embedded within the cannula hub itself, without bulkier external modules.
- Wireless, battery-free designs powered by near-field communication or body heat.
- Predictive AI models that estimate infiltration risk based on patient history, drug type, and insertion site before complications appear.
- Integration with smart infusion pumps that automatically pause infusion when infiltration risk crosses a threshold.
- Affordable smart cannulas for emerging markets — a critical step if the global infiltration burden is to be reduced meaningfully.
The trajectory is clear: IV therapy is moving from a passive delivery system to an active, intelligent one.
Conclusion: From Detection to Prevention
For a century, the IV cannula has been one of the most familiar, and most overlooked, devices in medicine. AI-powered smart cannulas promise to change that, transforming a routine procedure into a continuously monitored, intelligently managed therapy.
For hospitals, the case is clear: better detection means fewer injuries, lower costs, and stronger compliance. For manufacturers, it means rethinking what a “disposable” can do. For patients, it means a safer, less painful, and more dignified hospital experience.
The future of IV therapy will not be defined by sharper needles or smoother polymers alone. It will be defined by the intelligence built into every device, and by the manufacturers committed to bringing that intelligence to every hospital, in every market.
About Lars Medicare
Lars Medicare is a Delhi-based manufacturer and global exporter of medical disposables with over 25 years of experience. Our portfolio includes IV cannulas, safety IV cannulas, Blood Control Technology (BCT) devices, closed-loop IV cannulas, IV infusion sets, three-way stopcocks, hypodermic syringes, gloves, and respiratory devices. We supply trusted, certified medical devices to healthcare partners across more than 50 countries.
Get in touch to learn more about our IV cannula range or to discuss partnership opportunities for next-generation IV therapy products.